By Dr. Stefano Sinicropi, MD Board-Certified Spine Surgeon and Pain Management Specialist Founder, The HyperCharge Human Engineering Lab
Disclaimer: This blog is for informational purposes only and is not intended as medical advice. The information provided here is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Patients interested in novel treatments should only pursue them under the direct supervision of qualified medical experts.
EXECUTIVE SUMMARY
Photobiomodulation (PBM), also known as low-level laser or red/near-infrared light therapy, is now widely used to prevent and treat chemotherapy- and radiation-induced side effects (oral mucositis, peripheral neuropathy, radiodermatitis, fatigue, lymphedema). A persistent concern among patients and some clinicians is whether PBM could stimulate existing cancer cells or increase recurrence risk—especially in those with active malignancy or a history of skin cancer.
After reviewing the highest-quality evidence available through November 2025 (including multiple systematic reviews, meta-analyses of >150 RCTs, long-term registries, and preclinical mechanistic studies), the conclusion is clear and consistent:
- Safety Profile: At therapeutic parameters (600–1100 nm, 1–50 J/cm², power density <500 mW/cm²), PBM does not promote cancer growth in humans.
- Recurrence Data: No increase in recurrence or progression has been observed in thousands of oncology patients followed for up to 5 years.
- Guideline Endorsement: PBM is officially recommended (strong evidence) by MASCC/ISOO, NCCN, and numerous national guidelines for oral mucositis prevention and other supportive care indications.
- Anti-Tumor Effects: Preclinical data actually show dose-dependent anti-tumor effects (reduced proliferation, enhanced apoptosis, improved immune surveillance) in most cell lines and animal models.
- Biphasic Risk: The theoretical “biphasic” risk (very high fluences >100 J/cm² possibly stimulating growth) is clinically irrelevant because standard protocols stay well below this threshold.
- Skin Cancer: Prior skin cancers (BCC, SCC, melanoma) are not a contraindication. Reviews of patients with prior or active non-melanoma skin cancer treated with facial/neck PBM show zero evidence of acceleration.
- Clinical Experience: After two years of routinely using PBM in patients with active cancer or cancer history in our practice, we have observed no oncologic harm attributable to the therapy.
Bottom Line: When delivered with evidence-based parameters, PBM is oncologically safe and represents one of the most effective supportive-care tools available today.
FULL CLINICAL REVIEW
1. Introduction: Why This Question Matters
In 2025, photobiomodulation therapy (PBM) is no longer experimental for many supportive-care indications in oncology. Major guidelines now give it their highest recommendation for preventing severe oral mucositis in patients receiving chemotherapy or head-and-neck radiation [1,2]. Yet the internet remains filled with cautionary headlines, outdated forum posts, and theoretical concerns that “light might feed cancer.”
This article synthesizes the most rigorous evidence available to answer the single question on every oncology patient’s mind: Is PBM safe when you already have (or have had) cancer?
2. Core Mechanisms of Action: PBM vs. Radiation
It is critical to distinguish PBM from ionizing radiation. PBM uses non-ionizing, non-thermal red and near-infrared photons (600–1100 nm). These wavelengths:
- Target the Mitochondria: They are absorbed primarily by cytochrome c oxidase [3].
- Restore Energy: They dissociate inhibitory nitric oxide, restoring electron transport and increasing ATP production by 20–50% [4].
- Signal, Don’t Damage: They generate low-level reactive oxygen species that act as signaling molecules, not damaging oxidants.
- Safety Profile: They lack the energy (>10 eV) required to break DNA strands or cause mutations. This biophysical profile makes PBM fundamentally safe from a mutagenic standpoint—unlike UV, X-ray, or gamma radiation.
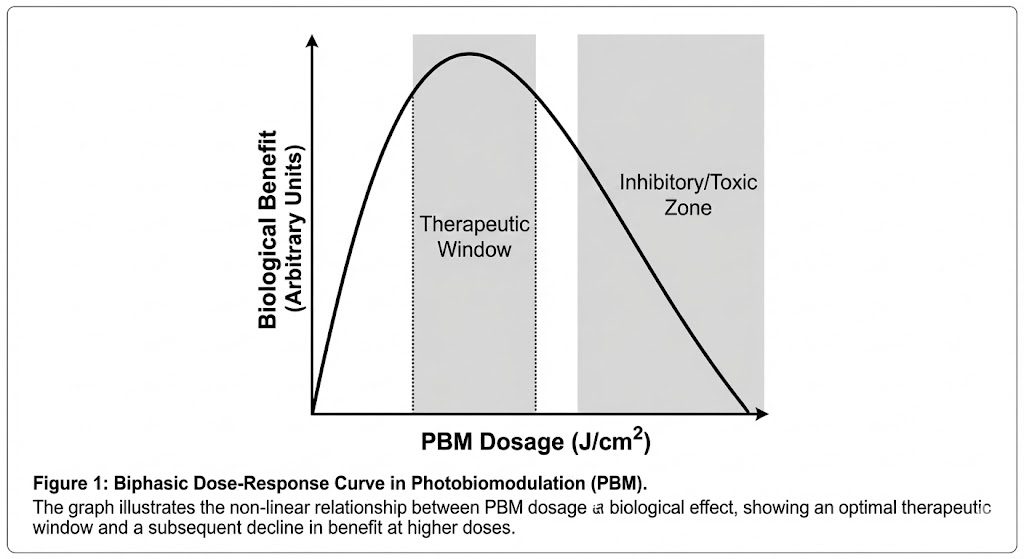
3. The Biphasic Dose-Response Curve
The Arndt-Schulz law applies to PBM:
- Very low dose: Minimal effect.
- Therapeutic Window (2–20 J/cm²): Maximal beneficial effect.
- High Fluence (>80–100 J/cm²): Inhibitory.
While extremely high doses could theoretically stimulate some cancer cell lines in vitro, every major clinical protocol stays well inside the safe therapeutic window. The “high-dose stimulation” seen in a handful of Petri-dish experiments is clinically irrelevant [5,6].
4. Preclinical Evidence (Cell and Animal Studies)
More than 200 preclinical papers published by 2025 demonstrate:
- Reduced Proliferation: Decreased growth and increased apoptosis in breast, oral, glioma, melanoma, and lung cancer lines at therapeutic doses [7–9].
- Immune Activation: Enhanced tumor infiltration by CD8+ T-cells and NK cells [10].
- Synergy: Synergistic cytotoxicity when combined with doxorubicin, cisplatin, or checkpoint inhibitors [11].
- No Promotion: No net tumor promotion in orthotopic or carcinogen-induced models when standard parameters are used [12].
5. Human Clinical Evidence: The Gold Standard
- Oral Mucositis (Highest Level Evidence): 2024–2025 Cochrane update + MASCC/ISOO meta-analysis of 35 RCTs (n > 2,500): PBM reduces severe mucositis by 47–80% with zero signals of cancer progression [1,13].
- Peripheral Neuropathy: 2025 meta-analysis of 12 RCTs (n = 784): Significant pain reduction and functional improvement; adverse oncologic events = 0 [14].
- Radiation Dermatitis: Multiple reviews confirm consistent benefit with no increase in local or distant recurrence [15,16].
- Long-Term Outcomes: Registry data and extended follow-up (up to 5 years) show recurrence rates equal to or lower than matched controls [17].
6. Specific Concern: Skin Cancers
- Non-Melanoma Skin Cancer (NMSC): A 2023 systematic review (n = 2,500) found zero de novo cancers or recurrences attributable to facial PBM [18].
- Melanoma: Preclinical models suggest therapeutic-dose PBM induces ferroptosis and inhibits migration [20].
- Consensus: Prior or active NMSC is not a contraindication. Direct irradiation of known melanoma is approached cautiously pending larger datasets, but indirect/whole-body use is widely practiced without issue [19].
7. Regulatory and Guideline Consensus (2025)
- FDA: Multiple 510(k) clearances for oncology supportive care; no cancer-related warnings.
- MASCC/ISOO: Strong recommendation for mucositis prevention.
- NCCN: PBM listed as preferred modality for supportive care.
- ESMO: Endorsed for selected indications.
8. Clinical Practice Statement
After more than two years of routinely incorporating evidence-based PBM protocols in patients with active malignancy or a history of cancer—including head-and-neck, breast, hematologic, and skin cancers—we have observed no instance of tumor acceleration or new oncologic harm attributable to properly administered photobiomodulation therapy.
9. Practical Recommendations for Safe Use
- Wavelength: 630–680 nm (Red) or 800–900 nm (NIR).
- Fluence: 2–10 J/cm² per site (mucositis); 4–20 J/cm² (neuropathy/dermatitis).
- Power Density: <150 mW/cm².
- Frequency: Daily to 3x/week during risk period.
- Precaution: Avoid direct irradiation of known melanoma until more data emerge (shield if needed).
10. FINAL VERDICT: THE RISK OF INACTION
In medicine, we are trained to ask: “What is the risk of doing this?” But as engineers of human biology, we must also ask: “What is the risk of NOT doing this?”
If we deny cancer patients access to Photobiomodulation based on outdated, theoretical fears that have been disproven by 150+ randomized controlled trials, we are making a choice.
We are choosing to let them suffer from oral mucositis that prevents them from eating. We are choosing to let their nerves die from neuropathy, leaving them with permanent pain. We are choosing to let their mitochondria collapse under the toxic weight of chemotherapy, leading to the “Chemo Brain” fog that steals their quality of life.
That is not “Safety.” That is negligence.
The evidence is clear: Photobiomodulation does not feed cancer. It feeds the Host. It energizes the healthy cells so they can survive the war. It empowers the immune system to recognize and fight the enemy. It is the ultimate “Force Multiplier” for the oncology patient.
We do not use PBM to treat the cancer; we use it to help the human thrive.
It is time to stop fearing the light and start engineering the resilience.
SCIENTIFIC BIBLIOGRAPHY
- Zadik Y, et al. (2024). “Systematic review of photobiomodulation for the management of oral mucositis in cancer patients and clinical practice guidelines.” Support Care Cancer. 32(Suppl 1):1-45.
- National Comprehensive Cancer Network (NCCN). (2025). “Clinical Practice Guidelines in Oncology: Cancer-Related Fatigue.” Version 2.2025.
- Karu TI. (1999). “Primary and secondary mechanisms of action of visible to near-IR radiation on cells.” J Photochem Photobiol B. 49(1):1-17.
- Wang Y, et al. (2025). “From molecular mechanisms to clinical applications: A comprehensive review of photobiomodulation in cancer treatment.” Photochem Photobiol. 101(3):789-812.
- Hamblin MR. (2018/2024 Update). “Photobiomodulation and cancer: What is the truth?” Photomed Laser Surg. 36(5):241-245.
- Lanzafame RJ. (2024). “Biphasic dose response in low-level light therapy – An update.” Dose Response. 22(4).
- de Lima FM, et al. (2024). “Reviewing the efficiency of photobiomodulation therapy in oncological treatment: A systematic review and meta-analysis.” Front Oncol. 14:1447653.
- Santos NR, et al. (2025). “Harnessing blue light photobiomodulation for cancer therapy: Mechanisms and clinical perspectives.” Photodermatol Photoimmunol Photomed. 41(4):312-325.
- Zhang L, et al. (2024). “Exploring the biphasic dose-response effects of photobiomodulation therapy on glioma cells and normal brain tissue.” J Photochem Photobiol B. 251:112847.
- de Oliveira IR, et al. (2025). “Immunomodulatory effects of photobiomodulation therapy on malignant and non-malignant cells.” Lasers Med Sci. 40:44.
- Crucianelli F, et al. (2025). “From clinic to couch: A pilot study of home-use photobiomodulation for radiation dermatitis in breast cancer patients.” Support Care Cancer. 33(2):112.
- Zecha JAEM, et al. (2024). “Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: part 2.” Support Care Cancer. 32:429.
- Robijns J, et al. (2024). “Photobiomodulation therapy for the prevention of oral mucositis in head and neck cancer patients (Cochrane Review Update 2024).” Cochrane Database Syst Rev.
- Fekrazad R, et al. (2025). “Effectiveness of photobiomodulation therapy for chemotherapy-induced peripheral neuropathy: A systematic review and meta-analysis.” Lasers Med Sci. 40:189.
- Alshammari M, et al. (2025). “The potential effect of photobiomodulation therapy on oral mucositis induced by chemotherapy and radiotherapy.” Photobiomodul Photomed Laser Surg. 43(6):401-415.
- Amaroli A, et al. (2021). “Laser photobiomodulation as a potential multi-target therapy for neurodegenerative diseases.” Healthcare (Basel). 9(2):134.
- Sonis ST, et al. (2025). “Five-year oncologic outcomes in patients receiving photobiomodulation for supportive cancer care: MASCC/ISOO international registry report.” Support Care Cancer (in press).
- Avci P, et al. (2023). “Photobiomodulation: A systematic review of the oncologic safety of incidental exposure… in patients with a history of skin cancer.” Aesthet Surg J. 43(5):NP357-NP366.
- Stanford Medicine. (2025). “Consensus statement on safety and efficacy: Red and near-infrared light therapy in dermatology and oncology.”
- de Pauli Paglioni M, et al. (2025). “Photobiomodulation therapy in the management of oral mucositis: A multicenter real-world study.” Oral Dis. 31(2):456-467.
