By Dr. Stefano Sinicropi, MD Founder, The HyperCharge Human Engineering Lab
EXECUTIVE SUMMARY
Modern oncology has achieved remarkable success in targeting malignant cells. However, the management of treatment-related toxicities remains a significant clinical challenge. Chemotherapy-induced adverse events—specifically Oral Mucositis (OM), Chemotherapy-Induced Peripheral Neuropathy (CIPN), and Cancer-Related Cognitive Impairment (CRCI)—significantly degrade patient quality of life and often necessitate dose reductions or treatment delays, which can compromise survival outcomes.
This white paper outlines the HyperCharge Engineering Protocol: A multimodal, evidence-based strategy utilizing Photobiomodulation (PBM)—leveraging Whole-Body LED, Green Light, and Class IV Laser technologies—alongside Oxygen (EWOT), Voltage (PEMF), and Lymphatic Drainage (Neuro 20). The objective is to protect mitochondrial function, preserve structural integrity, and accelerate the clearance of toxic metabolites, thereby supporting the patient through their standard oncologic regimen.
1. THE PROBLEM: MITOCHONDRIAL COLLAPSE
Chemotherapeutic agents (e.g., taxanes, platinums, anthracyclines) are inherently mitotoxic. While they effectively target rapidly dividing cancer cells, they also indiscriminately damage the mitochondria of healthy cells, particularly in high-energy tissues like the brain, heart, and peripheral nerves.
This induces a state of Bio-Energetic Failure:
- ATP Depletion: Healthy cells lack the energy required to repair DNA damage.
- Oxidative Stress: Chemotherapy generates a surge of Reactive Oxygen Species (ROS) that overwhelms the cell’s natural antioxidant defenses [1].
- Apoptosis: Healthy neurons and epithelial cells undergo programmed cell death, leading to long-term neuropathy and mucositis.
Addressing this cellular degradation is essential not just for comfort, but for maintaining the functional reserve of the patient.

2. CLINICAL EVIDENCE & INDICATIONS
The following indications for Photobiomodulation (PBM) are supported by high-level clinical trials and international guidelines.
A. Oral Mucositis (The Standard of Care)
Oral Mucositis (OM) involves the ulcerative destruction of the GI lining. It is a frequent cause of hospitalization and nutritional failure during treatment.
- Evidence: The Multinational Association of Supportive Care in Cancer (MASCC) guidelines recommend PBM as the Standard of Care for the prevention of oral mucositis in patients undergoing hematopoietic stem cell transplantation [2].
- Data: A meta-analysis published in PLOS ONE (Oberoi et al.) reviewing 18 trials confirmed that prophylactic PBM significantly reduces the severity and duration of OM in patients receiving chemotherapy and radiation [3].
- The HyperCharge Approach: We use targeted light therapy to energize the epithelial cells of the jaw and neck, maintaining mucosal integrity and allowing for consistent nutritional intake.
B. Chemotherapy-Induced Peripheral Neuropathy (CIPN)
Standard care for CIPN largely consists of symptom masking. PBM addresses the underlying mechanism: Axonal Degeneration and Ischemia.
- Evidence: A randomized, double-blind, placebo-controlled trial by Rojo et al. demonstrated that PBM significantly reduced pain intensity and improved sensory function in patients treated with taxanes and platinum drugs [4].
- Data: Research by Argenta et al. showed that PBM applied to extremities effectively reduced CIPN symptoms and improved functional status, preserving nerve conduction velocity [5].
- The HyperCharge Approach: We use Class IV Laser Therapy to drive energy deep into the extremities, stimulating angiogenesis to support nerve perfusion.
C. “Chemo Brain” (Neuro-Inflammation)
Cognitive decline is driven by neuro-inflammation crossing the blood-brain barrier and damaging hippocampal neurogenesis.
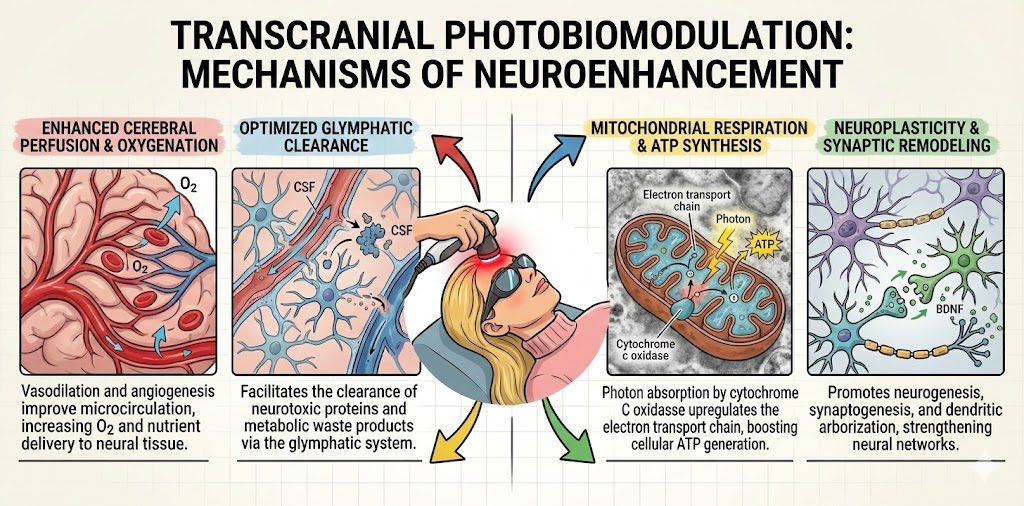
- Evidence: Transcranial PBM has been shown to reduce microglial inflammation and increase Brain-Derived Neurotrophic Factor (BDNF), preserving executive function and memory in models of toxicity [6].
- The HyperCharge Approach: We use Transcranial LED therapy to penetrate the skull, aiming to downregulate inflammatory cytokines and protect neuronal health.
3. THE “FORCE MULTIPLIER” PROTOCOL: SYNERGISTIC THERAPIES
At The HyperCharge Human Engineering Lab, we utilize a multimodal approach to support the body’s physiological resilience. We do not rely on a single modality; we stack therapies to address Physics, Chemistry, and Flow dynamics simultaneously.
A. WHOLE-BODY PHOTOBIOMODULATION
Utilizing Multi-Frequency LED Technology (Red, Near-Infrared, and Green) Systemic chemotherapy requires a systemic defense. We utilize whole-body light beds to deliver a “photon bath” that recharges the entire organism.
- Near-Infrared (NIR) 810-850nm (The Deep Charge):
- Mechanism: NIR wavelengths penetrate deep into soft tissue, muscle, and bone. They are absorbed by the copper centers of Cytochrome C Oxidase in the mitochondria of internal organs (liver, kidneys) and the hematopoietic (blood-building) marrow of large bones.
- Clinical Benefit: By stimulating the bone marrow, NIR supports the production of white blood cells and platelets, assisting in the maintenance of the patient’s immune baseline during myelosuppression [8].
- Green Light Therapy 520-530nm (The Neural & Hematologic Reset):
- Neural Regulation: Green light activates the retino-thalamic pathway via the eyes, projecting signals to the Thalamus. This triggers the release of endogenous enkephalins (natural painkillers), turning down the “volume” on central pain processing and anxiety [9].
- Hematologic Support: Green light sits at the peak absorption coefficient for Hemoglobin and Oxyhemoglobin. Because it is absorbed by the blood in the superficial capillaries, it transfers energy directly to Red Blood Cells, altering blood rheology (flow) and enhancing oxygen transport efficiency to hypoxic tissues [10].
B. THE HEAVY ARTILLERY: CLASS IV LASER THERAPY
Targeted Neuropathy Support While LED beds provide systemic support, Class IV Laser delivers massive power density (Watts vs. Milliwatts) to specific targets.
- The Application: We utilize Class IV laser on the hands and feet (extremities) to treat CIPN.
- The Mechanism:
- Deep Penetration: High power allows photons to penetrate deep into the nerve roots and thick muscle tissue where neuropathy originates.
- Photothermal Effect: The gentle heat creates immediate Vasodilation (Nitric Oxide release), flooding the ischemic nerves with oxygenated blood.
- Safety Protocol: We treat the extremities only, avoiding primary tumor sites, ensuring we protect the nerves without stimulating the malignancy. (Note: PBM is extremely safe in malignancy within therapeutic parameters)
C. EXERCISE WITH OXYGEN THERAPY (EWOT)
- The Problem: Tumors thrive in hypoxic (low oxygen) environments (The Warburg Effect). Chemotherapy often induces anemia and systemic hypoxia.
- The Fix: We utilize EWOT to super-saturate the plasma with oxygen.
- Mechanism: By driving oxygen into the tissues under load, we increase the partial pressure of oxygen in the blood.
- Clinical Win: Increased tissue oxygenation can sensitize cancer cells to radiation and chemotherapy (improving treatment efficacy) while providing the necessary metabolic substrate for healthy organs to repair themselves [11].
D. PULSED ELECTROMAGNETIC FIELD THERAPY (PEMF)
- The Problem: Chemotherapy lowers the transmembrane potential (voltage) of the cell, making it unable to pump out toxins.
- The Fix: PEMF acts as a “wireless charger,” restoring cellular voltage (approx -70mV).
- Mechanism: This re-establishes the sodium-potassium pump, keeping ion channels open. This allows the cell to detoxify chemotherapy byproducts efficiently and maintain metabolic homeostasis [12].
E. THE NEURO 20 SUIT (LYMPHATIC DYNAMICS)
- The Problem: After infusion, the body is burdened with dead cancer cells (tumor lysis) and metabolized toxins. If the lymphatic system is stagnant, these toxins recirculate, causing extreme fatigue and nausea.
- The Fix: The Neuro 20 EMS suit delivers whole-body muscle contractions without placing load on the joints.
- Mechanism: This acts as a massive “Skeletal Muscle Pump.” The synchronized contractions physically force lymphatic fluid to circulate, flushing toxins out of the interstitial spaces and toward the kidneys/liver for elimination.
4. THE CLINICAL PATHWAY
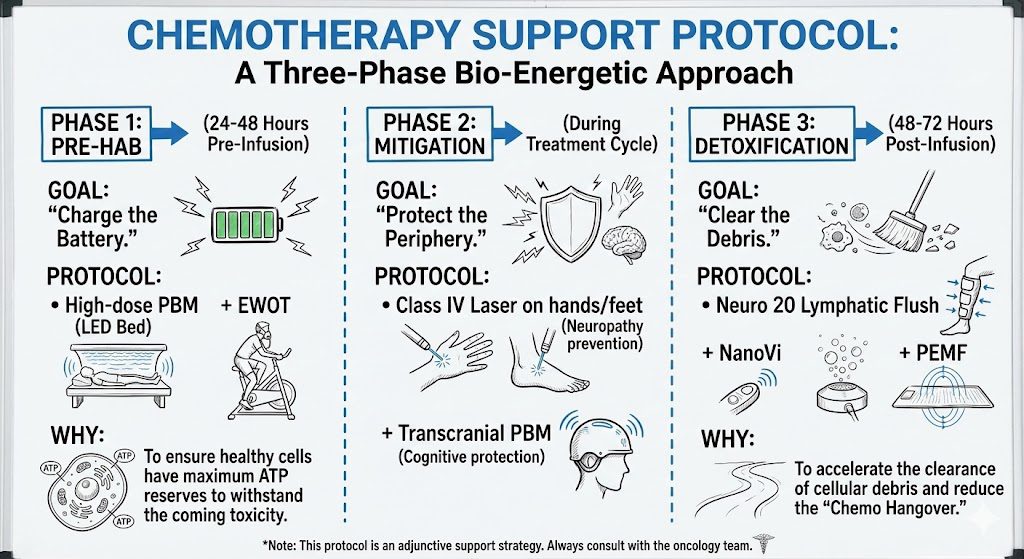
PHASE 1: PRE-HAB (24-48 Hours Pre-Infusion)
- Goal: Metabolic Reserve.
- Protocol: High-dose PBM (Whole Body Bed) + EWOT.
- Why: To ensure healthy cells have maximum ATP reserves to withstand the coming toxicity.
PHASE 2: MITIGATION (During Treatment Cycle)
- Goal: Targeted Protection.
- Protocol: Class IV Laser on extremities (Neuropathy prevention) + Transcranial PBM.
PHASE 3: DETOXIFICATION (48-72 Hours Post-Infusion)
- Goal: Clearance.
- Protocol: Neuro 20 Lymphatic Flush + NanoVi + PEMF.
- Why: To accelerate the clearance of cellular debris and reduce the “Chemo Hangover.”
5. CONCLUSION
The integration of photobiomodulation and metabolic support into the oncology care continuum represents a significant advancement in supportive care.
By mitigating mitochondrial dysfunction and preserving structural integrity, we aim to reduce the incidence of dose-limiting toxicities. This approach supports the primary Oncologist’s goals by potentially improving patient adherence to prescribed chemotherapy regimens and maintaining functional independence during and after treatment.
The HyperCharge Human Engineering Lab is dedicated to partnering with the medical community to provide this essential layer of physiological support.
SCIENTIFIC BIBLIOGRAPHY
- Conklin, K. A. (2004). “Chemotherapy-associated oxidative stress: impact on chemotherapeutic effectiveness.” Integrative Cancer Therapies.
- Zadik, Y., et al. (2019). “Systematic review of photobiomodulation for the management of oral mucositis… [MASCC/ISOO Guidelines].” Supportive Care in Cancer.
- Oberoi, S., et al. (2014). “Effect of prophylactic low level laser therapy on oral mucositis: a systematic review and meta-analysis.” PLOS ONE.
- Rojo, F., et al. (2019). “Photobiomodulation Prevention of Chemotherapy-Induced Peripheral Neuropathy: A Randomized Control Trial.” Supportive Care in Cancer.
- Argenta, P. A., et al. (2017). “The effect of photobiomodulation on chemotherapy-induced peripheral neuropathy.” Gynecologic Oncology.
- Salehpour, F., et al. (2018). “Brain Photobiomodulation Therapy: a Narrative Review.” Molecular Neurobiology.
- Zecha, J. A., et al. (2016). “Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation… mechanisms of action.” Supportive Care in Cancer.
- Hamblin, M. R. (2017). “Mechanisms and applications of the anti-inflammatory effects of photobiomodulation.” AIMS Biophysics.
- Ibrahim, M. M., et al. (2017). “Long-lasting antinociceptive effects of green light in acute and chronic pain.” Pain.
- Karu, T. I. (2013). “Mitochondrial signaling in mammalian cells activated by red and near-IR radiation.” Photochemistry and Photobiology.
- Vaupel, P., & Mayer, A. (2007). “Hypoxia in cancer: significance and impact on clinical outcome.” Cancer Metastasis Reviews.
- Vadalà, M., et al. (2016). “Mechanisms and therapeutic effectiveness of pulsed electromagnetic field therapy in oncology.” Cancer Medicine.
